Bioprinting: Creating New Milestones in Personalized Healthcare
Bioprinting is an additive manufacturing technology that utilizes a digital file as a blueprint to print objects layer by layer. Unlike traditional 3D printing, bioprinters use cells and biomaterials to produce organ-like structures that allow living cells to proliferate. Although bioprinting is a new technique, it has immense potential in advancing fields such as drug development, aesthetics, and regenerative medicine and personalized healthcare. Essentially, bioprinting is a type of 3D printing that can potentially produce anything from bone tissue and blood vessels to living tissues facilitating various medical applications, including tissue engineering and drug testing and development. The bioprinting process consists of three main steps:
- Pre-bioprinting: Anatomically accurate 3D models are created using computer graphics tools like CAD/CAM. These are later converted into a stack of 2D layers with user-definable thickness before being fed into the bioprinter for printing. The material or bio-ink selection is an integral part of this step. Digital files often based on MRI and CT scans are generated to enable the printer to interpret the data. Using a live-cell imaging device, researchers prepare the cells and combine them with bioink while ensuring that there are enough cells for effective bioprinting of a tissue model.
- Bioprinting: This bioprinting step involves the actual printing of tissues by additive manufacturing techniques. Depending on the structure researchers want to produce, the researchers load the cell-filled bio-ink into a cartridge and select one or more printheads. Different cell types, bioinks, and tools are used by researchers to create a variety of tissue forms.
- Post-bioprinting: This is the final stage of the bioprinting process which provides stability to the printed structure. Most structures require crosslinking to achieve complete stability. Crosslinking is typically accomplished by exposing the construct to either an ionic solution or UV light; depending on the composition of the construct. The constructs with cells are then placed inside an incubator for culture.
Bioinks in Bioprinting
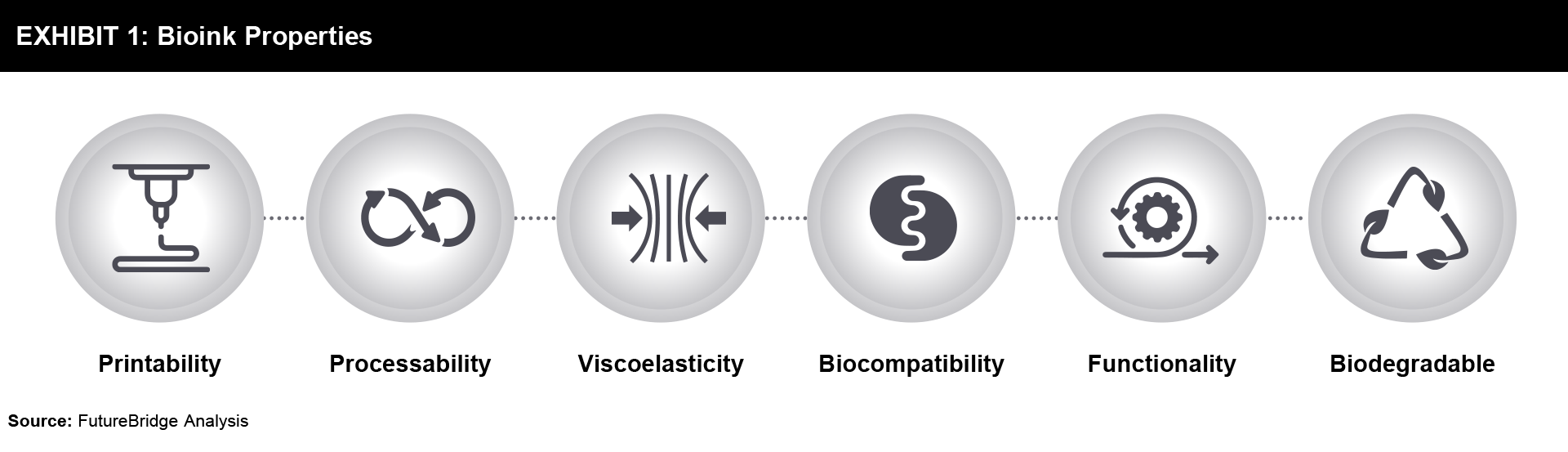
Biomaterials can be either synthetic or natural, or even a combination of both. In some cases, cell aggregates alone can serve as bioink for bioprinting procedures. Bioinks commonly consist of biomaterials such as hydrogels, cells, cell aggregates, or their combinations. Natural polymers like alginate and artificial polymers like gelatin are examples of materials used as bioinks. For the bioprinted tissues and organs to function properly, the ideal bioink must have the right mechanical, rheological, and biological qualities that can mimic the target tissues. Bioprintability, high mechanical integrity and stability, insolubility in cell culture media, biodegradability at a pace suited to the regenerating tissue, non-toxicity and non-immunogenicity, viscoelasticity, and the capacity to stimulate cell adhesion are few significant characteristics of an excellent bioink material. Additionally, bioink materials should be inexpensive, simple to produce and process, and readily available in the market. The ideal properties of bioink have been shown in Exhibit 1.

Applications of Bioprinting
Biocompatible materials are currently being developed for the biomedical industry offering significant advancements like the 4D bio-printing that possesses the form-changing properties of biomaterials and tissue-constructed structures. This innovative technology has diverse applications, including tissue engineering, drug delivery, and functioning organs. The impactful solutions bioprinting offers in healthcare and regenerative medicine include:
- Drug development: Much of today’s research relies on live subjects. Bioprinted tissue can be used in the preliminary stages instead of this and it also proves to be a more ethical and cost-effective solution. Using bioprinted tissue researchers can determine the efficacy of drug candidates earlier, saving money and time.
- Artificial organs: Lengthy organ waiting lists leave helpless patients waiting for years to get a suitable match that they need. The ability to bioprint organs could help doctors meet the demands of patients in need of organ transplants or even eliminate the need for transplantation altogether. The advanced bioprint organs solution offers many powerful options in this area.
- Wound healing: Many tissue-specific bioinks are now available, allowing researchers to work with artificial skin cells, neurons, liver cells, and more. In the future, these models could be used for therapeutic procedures such as skin grafts, bone dressings for war wounds, and even plastic surgery.
- Tissue engineering and regenerative medicine: Tissue engineering and regenerative medicine present significant challenges in the field of bioprinting functional organs, particularly in establishing vascular networks by connecting arteries, veins, and capillaries. The integration of diverse cell types to create complex tissue structures with mechanical and structural integrity has proven to be a challenging task. Despite these limitations, notable progress has been made in bioprinting thin or hollow tissues like blood vessels, as well as tissues such as cartilage that do not require vasculature. To create bioprinted cardiac tissue, tissue spheroids have been created from human vascular endothelial cells (HUVEC) and cardiac cells. Another crucial area of focus in tissue engineering is the fabrication of heart valves, which lack regenerative capabilities and therefore necessitate replacement with mechanical or biological prostheses upon damage. Bioprinting studies have demonstrated successful production of anatomically accurate axisymmetric aortic valve shapes.
- Cancer research: A major drawback of traditional two-dimensional tumor models are their limitations in representing the physiologically relevant environment required for understanding cancer pathogenesis and metastasis due to the absence of three-dimensional interactions with neighbouring cells and substrates. Bioprinting offers a platform for investigating cell interactions in a three-dimensional context enabling clinically relevant observations on cancer pathogenesis and metastasis. For instance, inkjet-based bioprinting has been utilized to bioprint human ovarian cancer cells (OVCAR-5) and MRC-5 fibroblasts. Additionally, framework-free bioprinting techniques of breast cancer models were demonstrated that encompass the physiologically relevant stromal environment, consisting of adipocytes differentiated from mesenchymal stem cells, breast fibroblasts, and endothelial cells.
Bioprinting Approaches
In general, bioprinting involves the precise layer-by-layer placement of biological components, biochemicals, and living cells to spatially control the arrangement of functional components within fabricated 3D structures. There are three fundamental approaches to bioprinting, which are summarized below:
- Biomimicry or Biomimetics: The first approach to bioprinting is called biomimicry which aims to create artificial structures that are identical to the natural structures found in human tissues and organs. Biomimicry involves replicating the shape, framework, and microenvironment of organs and tissues. The application of biomimicry in bioprinting involves creating both identical cellular and extracellular portions of organs. Tissues must be replicated at a microscale for this approach to be successful. Therefore, there is a need to understand the microenvironment, the nature of the biological forces within this microenvironment, the precise organization of functional and supportive cell types, solubility factors, and the composition of the extracellular matrix.
- Autonomous self-assembly: The second approach to bioprinting is autonomous self-assembly, which is inspired by the physical process of embryonic organ development as a model for replicating the tissue of interest. During the preliminary stages of development, cells create their own extracellular matrix building blocks, appropriate cell signaling, and independent assembly and patterning to provide the necessary biological functions and microarchitecture. Autonomous self-assembly requires in-depth understanding of embryonic tissue issue and organ developmental techniques. ‘Scaffold-less’ models use self-assembling spheroids, which undergo fusion and cell assembly to resemble developing tissues. Autonomous self-organization relies on cells as the fundamental drivers of tissue formation, controlling the building blocks, and structural and functional properties of these tissues. Achieving bioprinted tissues through this approach necessitates a deeper understanding of embryonic tissue mechanisms and the surrounding microenvironment.
- Mini-tissue: The third bioprinting approach is a combination of biomimicry and self-assembly approaches, referred to as mini-tissues. Mini-tissues are composed of minute functional parts of organs and tissues. The mini-tissue approach takes these small pieces, fabricates them, and places them in a larger frame. For example, the nephron serves as the mini tissue for constructing kidney tissues.
Challenges and available solutions in Bioprinting
With advancements in printing technology and the development of efficient and cost-effective printing methods, the need for stringent quality control standards throughout the bioprinting process has become necessary but challenging. One of the key challenges in bioprinting is ensuring quality control standards at each step of the process, including model design, bioink selection, printing validation, post-printing maturation, and product quality assessment. Regulating these standards can be difficult but is crucial for successful transplantation outcomes. Another significant challenge lies in the complexity of the bioprinting process, which involves multiple components. The lack of software to define the placements of cells, biomaterials, and biological molecules virtually following the robust designing and translation that drive downstream manufacturing operations hampers the bioprinting process. Some common challenges on the road to bioprinting are presented in Exhibit 2.
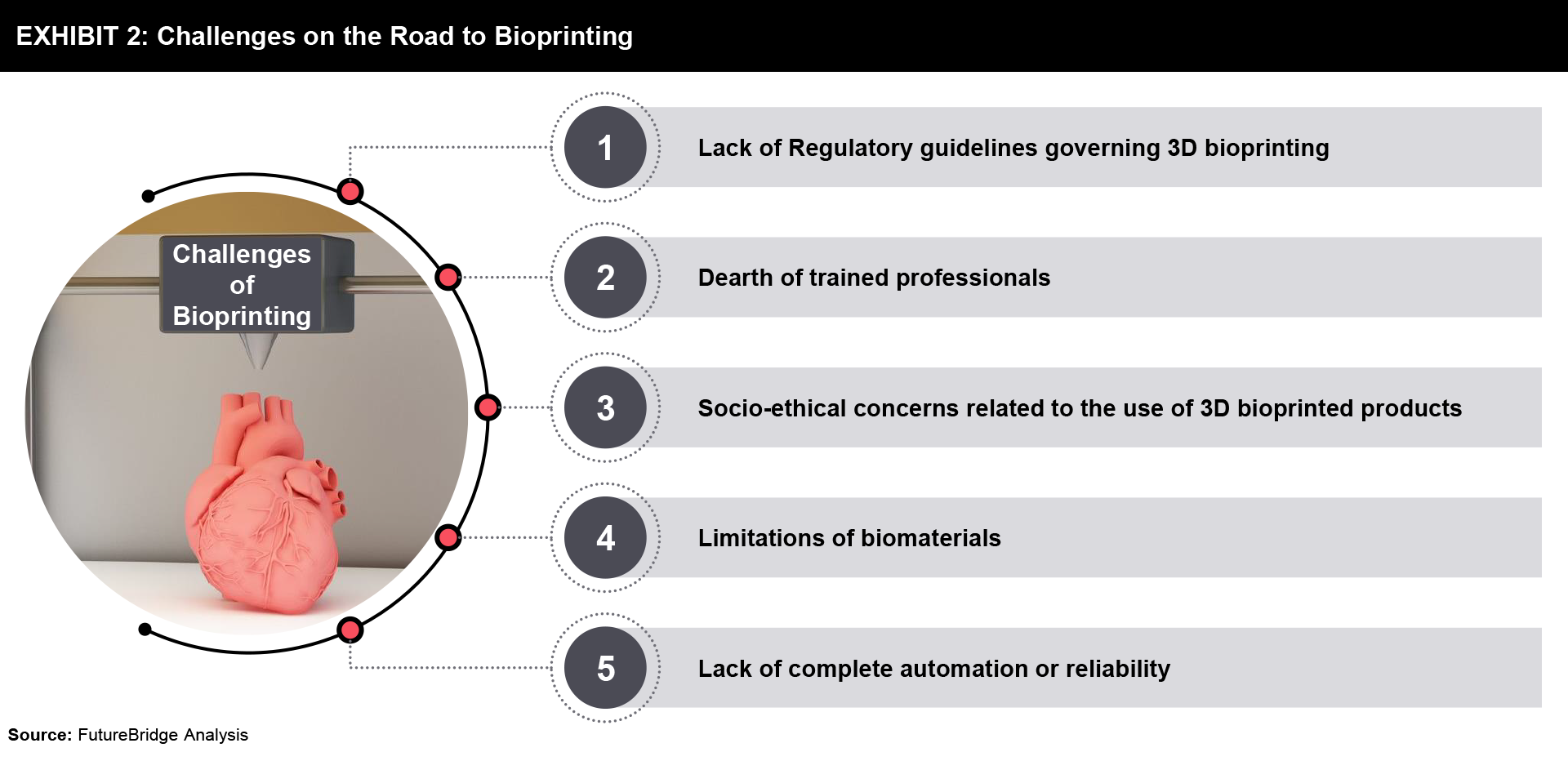

To address the challenges mentioned above, several solutions have been proposed and developed in the field of bioprinting.
Limited options for biomaterials:
- Supporting and providing structure to the injected cells that form functional printed organs is a challenging task for the required biomaterials. The biomaterials needed to support and give structure to the injected cells in functional printed organs face significant challenges. While synthetic polymers are mechanically robust, they often lack the necessary properties for good cell attachment. Conversely, natural polymers exhibit superior compatibility with cell adhesion, proliferation, and differentiation, despite not being as strong as their synthetic counterparts. As researchers continue ongoing studies for materials with just the right properties for bioprinting, avenues are opening to expand the range of sources for biomaterials. In a review article published in Frontiers in Mechanical Engineering, researchers from Swansea highlighted the potential of plant-based biomaterials in 3D tissue and organ printing. In particular, the team called attention to nanocellulose and alginate, pointing out that plant-based materials combine the power of plant microarchitecture with the inherent benefit of cell growth support.
Structure integrity:
- One challenge in bioprinting is the need to develop a 3D construct that is both mechanically rigid and stable for successful transplantation. The porosity and structure created during bioprinting should maintain a high elastic modulus to support natural cell proliferation. during implantation. If the scaffold is not maintained structurally and mechanically, the newly produced tissues may fail due to scaffold deformation. Maintaining structural integrity of the bioprinted tissue and seed cells can be challenging, especially depending on the viscosity of the bioink used. To retain the structure of bioprinted tissue and seed cells, biodegradable scaffolds composed of biomaterials are frequently utilized. However, they also come with certain disadvantages. They can induce an immune response, and there is a potential for toxicity and interference from degradation by-products that may affect cell-to-cell interactions.
- Researchers at the University of Birmingham have addressed this challenge by developing a unique method for printing soft biomaterials without letting them sag or losing their structure. The research team’s method of Suspended Layer Additive Manufacturing (SLAM) employs particles modified to provide self-healing characteristics to a hydrogel made of polymers. The gel’s protective properties allow cell-infused biomaterials to be injected into it and build up in layers to create a 3D shape, which ensures that the created structures are precise and without leaking or sagging.
Vascularization of organs:
- Bioprinting faces specific challenges related to internal vascularization of constructed organs. Creating circulatory networks within bioprinted organs has proven to be a formidable task. The compatibility of materials and cells with the printer and printing processes poses difficulties in printing a functional vascular network. Additionally, vascular networks may develop and mature at a slower rate compared to cells within the tissue structure.
- As an additional approach to producing functional vasculature for bioprinted organs, a research team at Harvard University’s Wyss Institute for Biologically Inspired Engineering has developed a new bioprinting technology termed SWIFT (Sacrificial Writing into Functional Tissue). To create a structure that resembles the cellular density of human organs, this technology inserts a network of vascular channels into a live matrix of stem cell-derived organ building blocks. This method also permits the administration of nutrients and oxygen through the vascular channels. By printing the ‘sacrificial ink’ into the matrix and shifting other cells out of the way without harming them, a thin bioprinting nozzle creates the channels.
Recent Advancements in Bioprinting
Bioprinting has made significant strides in recent years, leading to successful transplantation of printed organs.
- A Queens-based biotech business called 3DBio Therapeutics claimed to have created a body component using a patient’s own cells for the first time via 3-D printing. The new ear was precisely printed to match the woman’s left ear in shape. The company stated that the newly implanted ear, will continue to develop cartilage tissue, giving it the appearance and feel of a normal ear.
- Another remarkable achievement was seen at Toulouse University Hospital and the Claudius Regaud Institute, where a woman who lost her nose to cancer received a new one through bioprinting. A 3D-printed mould was implanted into her forearm to stimulate blood vessel growth, and after a two-month colonization period, it was successfully transplanted to the nasal region, allowing revascularization.
- In a four-year development effort, Mobius 3D Technology (M3DT) and the Netherlands Cancer Institute successfully transplanted a titanium lower jaw into a patient with head and neck cancer. The patient’s 3D MRI and CT scans were used to recreate a customized jawbone, which resulted in a flawless match for its new bony residence. Due to the custom-made nature of the implant, the jaw maintains its fit, and the pressure on the mucosa or skin above it is dispersed more evenly.
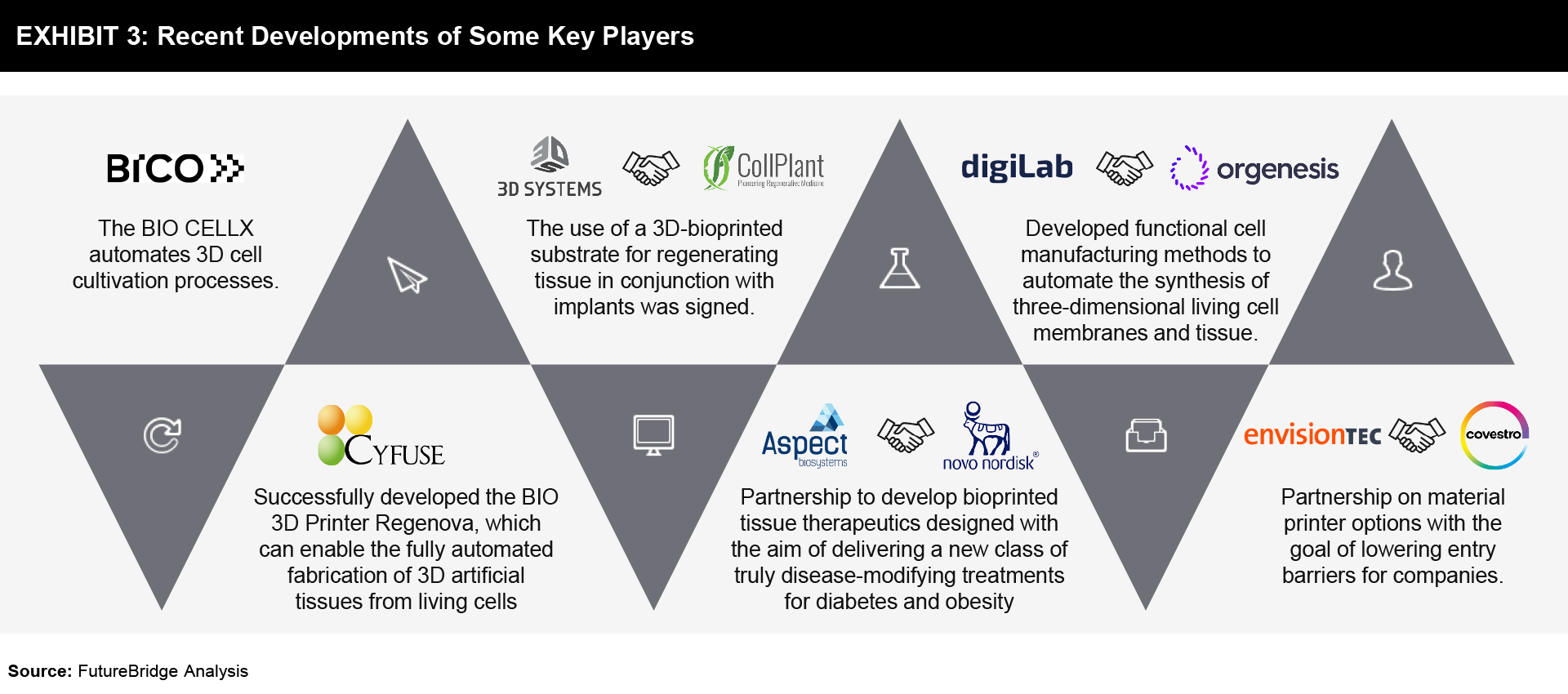
To achieve a competitive advantage, major players are concentrating on research and development to innovate new applications. For instance, CELLINK introduced the most sophisticated bioprinting tool, Bio X6 as well as Lumen X which specializes in creating vascular structures. Companies are joining forces and making acquisitions, partnerships, and mergers to increase their application portfolio, production capabilities, and competitive difference. For instance, BASF GmbH’s acquisition of the 3D printing company Sculpteo is expected to enhance their competitiveness and accelerate the development of new industrial bioprinting materials. The recent developments in bioprinting by select key players are shown in Exhibit 3.

Future Scope of Bioprinting
Bioprinting still has a long way to go before it can be used in a clinical context, especially in situ direct applications. Future research is now focusing on integrating different procedures to complement each other and improve the creation of tissue-mimicking structures. Applications for bioprinting, such as skin regeneration, cell and molecule delivery systems, and disease modelling, may be readily and easily available soon and enhance patient outcomes. These applications may transform how skin wound healing can occur faster. As bioprinting continues to progress, it will become increasingly feasible to print organ patches, complete replacement organs, as well as skin and bone grafts using a patient’s own cells. The use of bioprinting will empower medical professionals and researchers with the tools they need to better focus on therapies and enhance patient outcomes as personalized and regenerative medicine gain popularity.
In the future, key areas of research will include the development of novel bioprinting materials with superior biocompatibility, improved printability, and suitable mechanical properties. Focus on the creation of heterogeneous and gradient composite materials, the creation of biomaterials suitable for in situ bioprinting, the enhancement of the biological properties of materials using biomimetics or recombination with bioactive factors, and more will also accelerate. As the range of printing materials expands and their performance improves, bioprinting is expected to bring significant advancements to the field of healthcare technology.
Conclusion
Tissue and organ regeneration through bioprinting holds immense promise for additive manufacturing. It enables the creation of physiologically comparable tissue that gives patients better and more reliable functional results. Bioprinting techniques offer high-throughput tissue printing with precise spatial control and accurate cell patterning. In the US alone, there are over 120,000 people on organ waiting lists, and many more suffer from chronic conditions because of the long-term negative effects of post-transplant immunosuppression. Bioprinting is a revolutionary recent technology that has the potential to end the waiting list for organ transplants. Because bioprinting develops tissue from the ground up, there is less chance of immunological graft rejections, which help to alleviate the problems associated with donor shortage.
In the production of medicinal drugs, bioprinting offers provides a faster, cost-effective, and biologically relevant alternative to animal testing for drug efficacy evaluation. The field of medicine has seen major advancements because of bioprinting, including enhanced drug delivery systems and sugar stents for easier vein anastomosis during surgeries. Although bioprinting is still in its developmental stage and faces significant challenges, especially in terms of in situ applications, it is a rapidly evolving field with the potential to revolutionize modern medicine and healthcare. By overcoming hurdles and advancing bioprinting technologies, we can envision a future healthcare landscape where customized tissues, organs, and pharmaceuticals are readily available, enhancing patient well-being.
To gain deep insights into bioprinting and create new milestones for your brand connect with FutureBridge consultants today.
References
- https://nextbiglab.com/2021/12/10/bioprinting-advancements/
- https://www.news-medical.net/health/What-is-Bioprinting.aspx
- https://www.cellink.com/blog/bioprinting-explained-simply/
- https://www.frontiersin.org/articles/10.3389/fmech.2020.589171/full
- https://thebiologynotes.com/3d-bioprinting/
- https://academic.oup.com/ejcts/article/46/3/342/2755268
- https://all3dp.com/2/what-is-3d-bioprinting-simply-explained/
- https://www.sciencedirect.com/science/article/abs/pii/B9780323854306000066
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6533576/
- https://www.news-medical.net/life-sciences/Bioprinting-Applications.aspx
- https://onlinelibrary.wiley.com/doi/full/10.1002/mco2.194




































