The GLP-1 Body Reset: Why the Beauty Industry’s Window to Act Is Rapidly Closing
100M+
Projected global GLP-1 users by 2030, from fewer than 15M today

22%
Average body weight reduction on tirzepatide over 72 weeks (SURMOUNT-1, NEJM)

40%
Of weight lost on GLP-1 can be lean mass, removing the collagen substrate skin depends on
18 mo
Window for the beauty and dermocosmetic industry before GLP-1 skin and body care categories are defined and formulation standards are set
GLP-1 is creating a new skin baseline. Beauty is still formulating for the past.
Glucagon-like peptide-1 receptor agonists are not weight loss drugs with cosmetic side effects. They are systemic metabolic interventions that simultaneously alter adipose architecture, collagen substrate availability, hair follicle cycling, sebaceous gland activity, gut microbiome composition, and the neural reward pathways that drive beauty product consumption.
This brief makes one central argument: the companies that define the formulation science, clinical evidence, and category positioning for GLP-1 consumers now will hold leadership for a decade. Those that wait will enter a category already defined, clinically validated, and priced at premium by others.
Exhibit 01
The metabolic skin age paradox – Biologically younger. Structurally older.
GLP-1 therapy simultaneously improves systemic metabolic markers and degrades the structural conditions that govern skin integrity. No existing formulation addresses the gap between these two trajectories.
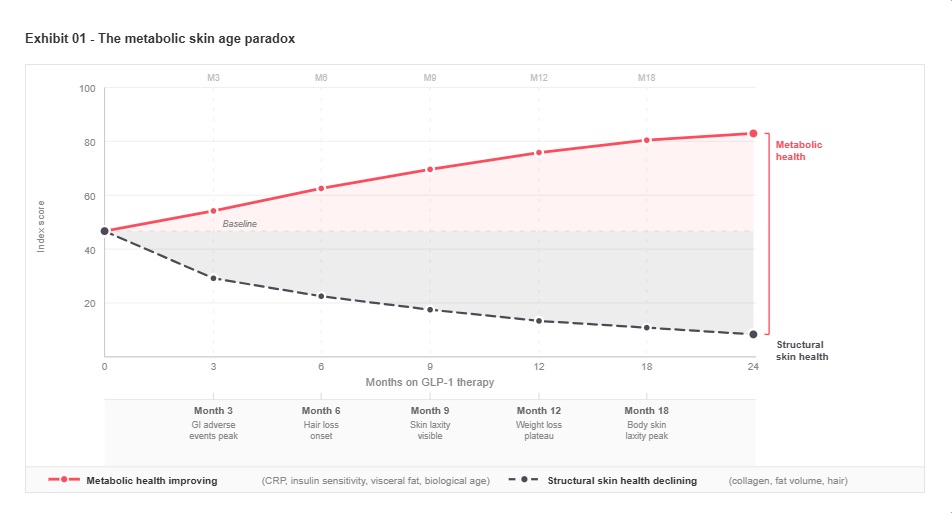
No current beauty or dermocosmetic formulation addresses the structural and metabolic changes induced by GLP-1 treatment
GLP-1 therapy is creating a new consumer paradox: individuals who are biologically younger by metabolic measures, yet structurally older in appearance. As metabolic health improves, reflected in lower inflammatory markers, restored insulin sensitivity, and reduced visceral adiposity, the simultaneous loss of subcutaneous fat removes critical structural support from the skin, altering dermal integrity and visible aging trajectories.
C-reactive protein levels fall by up to 43%, biological age improves by standard biomarker panels, and cardiovascular risk reduces measurably. In the same patient, subcutaneous fat supporting dermal architecture is lost at rates corresponding to 15 to 22% of total body weight over 12 months. The consumer looks structurally older in the mirror at the precise moment her physician is reading improving metabolic results. This divergence is unaddressed by the beauty industry. It is also, by definition, the commercial opportunity.
The loss of subcutaneous fat silences a critical collagen-signaling system, and no available formulation compensates for its absence
The beauty industry’s understanding of the “Ozempic face” phenomenon is largely mechanical: fat leaves, skin sags. This interpretation misses the underlying biology. Subcutaneous fat adipocytes produce adiponectin, a hormone that directly stimulates dermal fibroblast activity and collagen type I synthesis. When this fat depot is rapidly depleted, the local collagen-stimulating signal disappears alongside the volume. The consequence is not mechanical volume loss alone but biochemical suppression of the skin’s endogenous repair and renewal system.
GLP-1 receptors have additionally been identified in dermal fibroblasts and keratinocytes, meaning the drug acts directly on the biology of cells responsible for skin architecture, barrier function, and renewal. The anti-inflammatory downstream effects demonstrably improve psoriasis and hidradenitis suppurativa. The same receptor activation may also alter the controlled inflammatory signaling required for normal collagen turnover and epidermal differentiation. No topical formulation currently available compensates for either mechanism. This is the primary formulation gap and the first defensible intellectual property territory.

Subcutaneous fat is not a cosmetic feature. It is an active endocrine organ that drives dermal collagen synthesis. GLP-1 does not simply remove fat. It withdraws the hormonal signal that instructs skin to repair and renew itself.
GLP-1-driven gut disruption creates a systemic skin barrier opportunity
GLP-1 drugs alter gut motility and microbiome composition profoundly. The downstream consequence, which is less visible and entirely unaddressed by current beauty formulations, is significant restructuring of gut microbiome populations that reduce production of short-chain fatty acids. SCFAs are essential regulators of skin barrier function, transepidermal water loss, and cutaneous immune competence. The gut-skin axis disruption created by GLP-1 therapy generates a systemic skin barrier vulnerability that extends far beyond the face. Several of the world’s largest personal care conglomerates maintain documented microbiome research programs and have published peer-reviewed work on skin barrier science. None have redirected this capability toward the GLP-1 context publicly.
The suppression of food reward creates a durable behavioral shift toward beauty ritual
GLP-1 drugs reduce food cravings by modulating dopamine reward pathways in the brain. When a primary pleasure source is chronically reduced, humans reliably transfer hedonic investment to adjacent categories. Beauty rituals are among the most accessible, socially sanctioned, and immediately available hedonic experiences available to adults. GLP-1 users who were previously food-reward-dominant consumers may become beauty-ritual-dominant consumers.
This demand-creation effect is supported by historical behavioral analogy. Beauty expenditure increased measurably during COVID-19 lockdowns when food as a reward mechanism was eliminated. GLP-1 creates a personal, chronic, and prescribed version of the same constraint. The brands that position themselves at this intersection, prioritising sensory richness, ritual depth, and experiential value alongside functional efficacy, will capture a behaviorally motivated premium that has no current formulation incumbent
Exhibit 02
The complete GLP-1 patient journey – every phase is a formulation opportunity
The commercial opportunity spans the full treatment lifecycle including withdrawal, cooling, and re-initiation phases that no beauty brand has mapped. Comorbidities present additional intervention points across the entire arc.

Withdrawal and re-initiation create a cyclical skin stress pattern that no beauty brand has yet modelled
The majority of GLP-1 users discontinue treatment within two years, with studies showing that more than 60% regain weight after stopping. This discontinuation-and-restart cycle, increasingly common as prescribers and patients learn to manage tolerability, creates a pattern of repeated structural skin stress that compounds over time. Each withdrawal phase brings rapid fat redeposition to altered anatomical sites, each re-initiation phase repeats the adiponectin withdrawal sequence, and the cumulative structural skin debt increases with each cycle. The beauty industry has no product category for this patient. The pharmaceutical industry has documented the biology. The commercial gap is defined and addressable.
Hair loss follows a predictable timeline that is currently being missed by every major hair care group
Telogen effluvium from rapid caloric restriction is well-documented in dermatological literature. The anagen-to-telogen phase shift in hair follicles begins 3 to 6 months after the metabolic stress of significant weight reduction, making it predictable from the moment of prescription. GLP-1 receptors have additionally been identified in dermal papilla cells of hair follicles, suggesting direct drug-biology effects on hair cycle beyond the secondary effects of nutritional stress. Hair care brands that deliver targeted intervention at months 2 to 4, with clinical evidence specific to this context, would intercept consumers at the precise moment of maximum biological need and maximum emotional distress. The leading professional and mass-market hair care brands operated by global personal care groups have the brand equity, scientific infrastructure, and distribution to occupy this position. None has yet produced a clinically positioned GLP-1 companion hair product.
The new skin archetype requires new formulation science, not product line extensions
Consumers emerging from GLP-1 treatment represent a skin type that does not appear in any current dermatological classification. Their skin simultaneously presents an improved glycation profile, reduced inflammatory load, significantly depleted subcutaneous mechanical support, altered sebum patterns, suppressed collagen substrate signaling from adiponectin withdrawal, and potentially compromised barrier function from gut microbiome disruption. This is metabolically transformed skin, and it requires a new formulation category with dedicated clinical evidence, specific active combinations, and regulatory positioning that distinguishes it from existing skincare segments.
The companies that define this category in clinical and dermatological literature before academic consensus forms will own the category name, the consumer perception, and the retailer shelf position. The window for category creation leadership is 12 to 18 months. After that, the academic literature will define the category on terms that may not favour any single commercial player.
Exhibit 03
Strategic routes to GLP-1 category leadership
Three routes to market, mapped against speed of deployment and depth of scientific differentiation. Each route connects to a distinct biological pathway. The combination of routes defines the degree of category ownership.
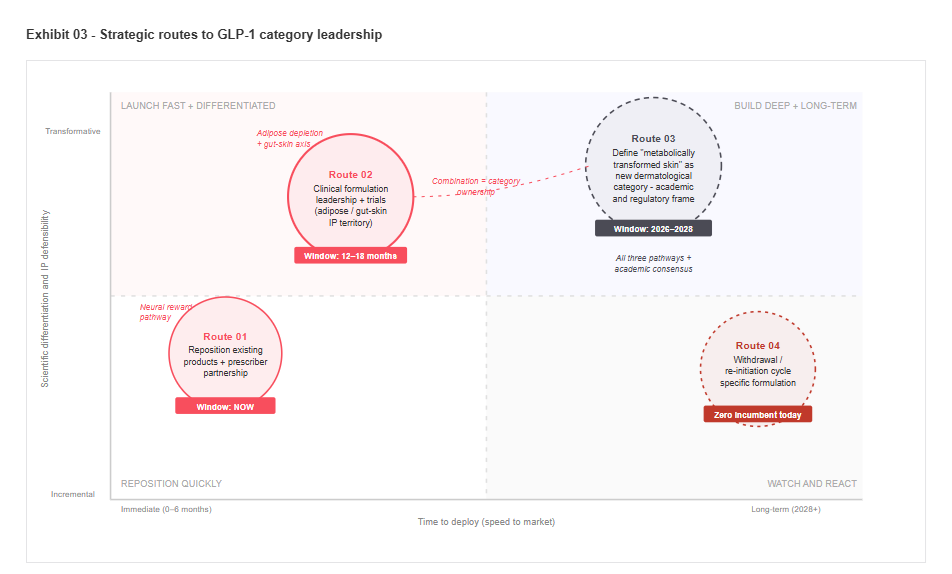
Three strategic routes are available. The window for two of them is already closing.
The companies positioned to capture value from the GLP-1 consumer transformation have three primary routes, each connecting to a distinct biological pathway and competitive position. The route chosen must match existing capability. The sequence of execution matters as much as the strategic choice.
01
Prescriber partnership and product repositioning
Negotiate companion protocol status with Novo Nordisk and Eli Lilly, positioning specific products as clinical recommendations at the point of GLP-1 prescribing. Simultaneously reposition existing barrier and nutritional products with GLP-1-specific clinical framing. The La Roche-Posay oncology programme demonstrates the model. GLP-1 represents that model at orders-of-magnitude greater patient volume.
Window: now.
02
Clinical formulation leadership and IP
Invest in GLP-1 dermatology R&D targeting the adipose depletion and gut-skin axis pathways. Generate clinical evidence for specific formulation categories and file IP on adiponectin-pathway activators, postbiotic gut-skin systems, and anagen-support scalp formulations. The active cosmetics and dermatological divisions of the largest personal care groups are the most capable incumbents.
Window: 12 to 18 months before academic consensus closes the first-mover advantage.
03
Category creation and definition
Define metabolically transformed skin as a new dermatological category in partnership with academic dermatology societies. Establish clinical practice frameworks that reference specific product standards. This route has the narrowest window. A major player that moves in the next 12 months can set the terms. One that waits will enter a category defined on someone else’s terms, with someone else’s clinical evidence.
The decision this brief is designed to accelerate
The GLP-1 consumer cohort is growing faster than any beauty company’s current R&D cycle. The biology is established in peer-reviewed literature. The patient journey is predictable, including withdrawal and re-initiation phases that no incumbent has formulated for. The formulation requirements are defined by existing dermatology, hair biology, and microbiome science. The gap between what the science requires and what the market currently provides is the commercial opportunity.
The companies that treat this as a strategic priority today will be entering clinical trials and pharmaceutical partnership discussions in 2026, with category-defining products reaching market in 2027. Those that classify it as a product adjacency will enter a category that others have defined, clinically validated, priced at premium, and distributed through prescription-adjacent channels they do not yet occupy.

GLP-1 starts with weight loss. Its actual consequence is the creation of a 100-million-person consumer cohort whose skin biology, hair cycle, gut microbiome, and behavioural relationship with beauty products have been fundamentally and predictably restructured by a single pharmaceutical class. The formulation response to that restructuring does not yet exist.

Author
Manoj Jiandani
Practice Head
Life Sciences, Home and Personal Care
Request A Confidential Discussion
How FutureBridge helps leadership teams act
In GLP-1–driven beauty disruption, the challenge is not identifying change. It is prioritizing where it matters first and acting before the category is defined.
We support leadership teams in identifying exposure across formulation pipelines, clinical positioning, ingredient strategies, and portfolio relevance. We assess where assumptions are no longer holding across collagen biology, skin barrier function, microbiome dynamics, and demand shifts driven by rapid weight loss and behavioral change. We benchmark competitive positioning beyond surface signals.
We translate this into clear decisions of where to recalibrate R&D focus, re-sequence innovation pipelines, and protect or redeploy capital. Most importantly, we help distinguish short-term aesthetic effects from structural biological shifts, so decisions are taken at the right time, on the right issues.




































